5.4
pH
What Is pH and why is it important?
pH is a term used to indicate the alkalinity or acidity of a substance as ranked on a
scale from 1.0 to 14.0. Acidity increases as the pH gets lower. Fig. 5.9 present the
pH of some common liquids.
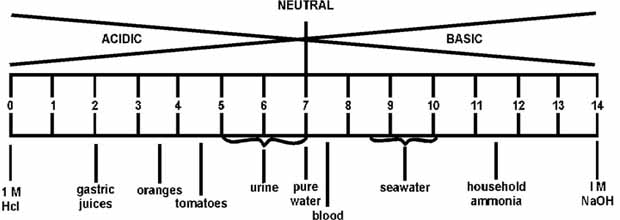
Figure 5.9
pH of selected liquids
|
pH affects many chemical and biological processes in the water. For example,
different organisms flourish within different ranges of pH. The largest variety of
aquatic animals prefer a range of 6.5-8.0. pH outside this range reduces the diversity
in the stream because it stresses the physiological systems of most organisms and
can reduce reproduction. Low pH can also allow toxic elements and compounds to
become mobile and "available" for uptake by aquatic plants and animals. This can
produce conditions that are toxic to aquatic life, particularly to sensitive species like
rainbow trout. Changes in acidity can be caused by atmospheric deposition (acid
rain), surrounding rock, and certain wastewater discharges.
The pH scale measures the logarithmic concentration of
hydrogen (H+) and hydroxide (OH-) ions, which make up
water (H+ + OH- = H2O). When both types of ions are in
equal concentration, the pH is 7.0 or neutral. Below 7.0, the
water is acidic (there are more hydrogen ions than hydroxide
ions). When the pH is above 7.0, the water is alkaline, or
basic (there are more hydroxide ions than hydrogen ions).
Since the scale is logarithmic, a drop in the pH by 1.0
unit is equivalent to a 10-fold increase in acidity. So, a
water sample with a pH of 5.0 is 10 times as acidic as
one with a pH of 6.0, and pH 4.0 is 100 times as acidic
as pH 6.0.
Analytical and equipment considerations
pH can be analyzed in the field or in the lab. If it is
analyzed in the lab, you must measure the pH within 2
hours of the sample collection. This is because the pH
will change due to the carbon dioxide from the air
dissolving in the water, which will bring the pH toward 7.
If your program requires a high degree of accuracy
and precision in pH results, the pH should be
measured with a laboratory quality pH meter and
electrode. Meters of this quality range in cost from around $250
to $1,000. Color comparators and pH "pocket pals" are suitable for
most other purposes. The cost
of either of these is in the $50
range. The lower cost of the
alternatives might be attractive if
you are relying on several teams
of volunteers sampling multiple
sites at the same time.
pH Meters
A pH meter measures the
electric potential (millivolts)
across an electrode when
immersed in water. This electric
potential is a function of the
hydrogen ion activity in the
sample. Therefore, pH meters
can display results in either
millivolts (mV) or pH units.
A pH meter consists of a
potentiometer, which measures
electric current; a glass
electrode, which senses the
electric potential where it meets
the water sample; a reference
electrode, which provides a
constant electric potential; and a
temperature compensating
device, which adjusts the
readings according to the
temperature of the sample
(since pH varies with
temperature). The reference and
glass electrodes are frequently
combined into a single probe
called a combination
electrode.
There is a wide variety of
meters, but the most important
part of the pH meter is the
electrode. Buy a good, reliable
electrode and follow the
manufacturer's instructions for
proper maintenance.
Infrequently used or improperly
maintained electrodes are
subject to corrosion, which
makes them highly inaccurate.
pH "Pocket Pals" and
Color Comparators
pH "pocket pals" are electronic
hand-held "pens" that are
dipped in the water and provide
a digital readout of the pH.
They can be calibrated to one
pH buffer (lab meters, on the
other hand, can be calibrated to
two or more buffer solutions
and thus are more accurate over
a wide range of pH
measurements).
Color comparators involve
adding a reagent to the sample
that colors the sample water. The intensity of the color is proportional to the pH
of the sample. This color is then matched against a standard color chart. The
color chart equates particular colors to associated pH values. The pH can be
determined by matching the colors from the chart to the color of the sample.
How to collect and analyze samples
The field procedures for collecting and analyzing samples for pH consist of
the following tasks.
TASK 1 Prepare the sample containers
Sample containers (and all glassware used in this procedure) must be
cleaned and rinsed before the first run and after each sampling run by
following the procedure described under Method A on page 128. Remember to
wear latex gloves.
TASK 2 Prepare before leaving for the sampling site
Refer to Section 2.3 - Saftey Considerations for details on confirming sampling date and time,
picking up and checking supplies, and checking weather and directions. In
addition to the standard sampling equipment and apparel, when sampling for pH,
include the following equipment:
- pH meter with combination temperature and reference electrode, or pH
"pocket pal" or color comparator
- Wash bottle with deionized water to rinse pH meter electrode (if
appropriate)
- Data sheet for pH to record results
Before you leave for the sampling site, be sure to calibrate the pH meter or
"pocket pal." The pH meter and "pocket pal" should be calibrated prior to
sample analysis and after every 25 samples according to the instructions that
come with them.
If you are using a "pocket pal," use the buffer
recommended by the manufacturer. If you are using a laboratory grade meter, use two pH
standard buffer solutions: 4.01 and 7.0. (Buffers can be purchased from test kit supply
companies, such as Hach or LaMotte.) Following are notes regarding buffers.
- The buffer solutions should be at room temperature when you calibrate the meter.
- Do not use a buffer after its expiration date.
- Always cap the buffers during storage to prevent contamination.
- Because buffer pH values change with temperature, the meter must have a built-in temperature
sensor that automatically standardizes the pH when the meter is calibrated.
- Do not reuse buffer solutions!
TASK 3 Collect the sample
Refer to Task 2 in Chapter 5 - Water Quality Conditions for details on how to collect water samples using screw-cap bottles or
Whirl-pak® bags.
TASK 4 Measure pH
The procedure for measuring pH is the same whether it is conducted in the field or lab.
If you are using a "pocket pal" or color comparator, follow the manufacturer's instructions. Use
the following steps to determine the pH of your sample if you are using a meter.
- Rinse the electrode well with deionized water.
- Place the pH meter or electrode into the sample. Depress the dispenser button once to
dispense electrolyte. Read and record the temperature and pH in the appropriate column on the
data sheet. Rinse the electrode well with deionized water. 3. Measure the pH of
the 4.01 and 7.0 buffers periodically to ensure that the meter is
not drifting off calibration. If it has drifted, recalibrate it.
TASK 4 Return the field data sheets and samples
to the lab or drop-off point.
Samples for pH must be analyzed within 2 hours of
collection. If the samples cannot be analyzed in the field, keep
the samples on ice and take them to the lab or drop-off point
as soon as possible within the 2-hour limit.
References
APHA. 1992. Standard methods for the examination
of water and wastewater. 18th ed. American Public
Health Association, Washington, DC.
River Watch Network. 1992. Total alkalinity and pH field and
laboratory procedures (based on University of Massachusetts
Acid Rain Monitoring Project). July 1.
< Previous · Table of Contents · Next >
Office of Wetlands, Oceans & Watersheds Home
Watershed Protection Home | Monitoring Water Quality Home
EPA Home |
Office of Water |
Search |
Comments


